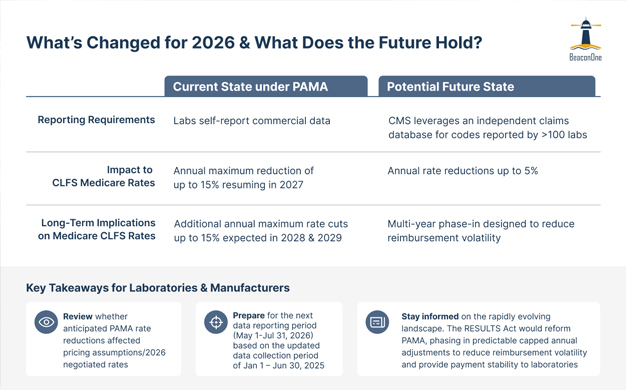
After much anticipation, the laboratory community has experienced a brief reprieve with the extended moratorium to Medicare CLFS rate reductions and provisions regarding the reporting requirements under PAMA. The decision to delay the 15% Medicare payment reduction through December 31, 2026, follows a prior five-year delay in PAMA-related payment reductions from 2021 through 2025. In addition to providing temporary payment relief, the legislation enacted under the Consolidated Appropriations Act, 2026 allows more time for Congress to enact permanent legislative reforms such as the RESULTS Act, which seeks to modernize the CLFS under PAMA. ACLA is advocating for the RESULTS Act to be included in an end-of-year (2026) spending package, which, if enacted, could lead to additional reforms to PAMA in 2027. With the rapid changes to PAMA that occurred in early 2026 and potential passage of the RESULTS Act, laboratories and manufacturers should closely monitor developments in the reimbursement and policy landscape for diagnostic tests, especially given the associated contractual and financial implications. BeaconOne Healthcare Partners is actively tracking and engaging in the evolving US and O-US policy and reimbursement landscape for laboratory services. Contact us to learn more.